Agentic Assistant
Clinical Intelligence.
One Conversation Away.
Answer protocol questions, surface operational metrics, and query live clinical data through natural language – directly inside your EDC.
Why Agentic Assistant?
The Intelligence Your Trials Deserve
Clinion is trusted by sponsors and CROs running clinical trials across 20+ countries. Agentic Assistant brings AI-powered intelligence to every conversation – unified, native, and ready from day one.
Unified Intelligence
Protocol intelligence, operational insights, and clinical data querying unified in a single conversational interface. No more fragmented tools, siloed dashboards, or cross-team dependencies.
Native EDC Integration
Embedded directly inside the Clinion EDC. No middleware, no connectors, no data sync delays. Real-time answers from the same source of truth your team enters data into every day.

Zero-Setup AI
Auto-generates a study-specific knowledge base the moment your study is created – indexing protocols, CRF structures, and operational metadata. Zero configuration. Zero white space between study start and AI readiness.
How it Works?
Four Steps. One Conversation.
Ask
Type your question in natural language. No query syntax. No special commands. Just ask the way you'd ask a colleague.
Classify
Agentic Assistant's intent classifier determines whether your question is about protocol knowledge, operational metrics, or clinical data.
Route
The question is routed to the specialized agent - Protocol Intelligence, Operations Insights, or Clinical Data, each with its own RAG pipeline and data access layer.
Respond
You receive a clear, formatted answer with traceable sources. Export to Excel or save as an image when you need to share it.
Explore
Explore Agentic Assistant
Features
Key Features of Clinical Trial Management Software (CTMS
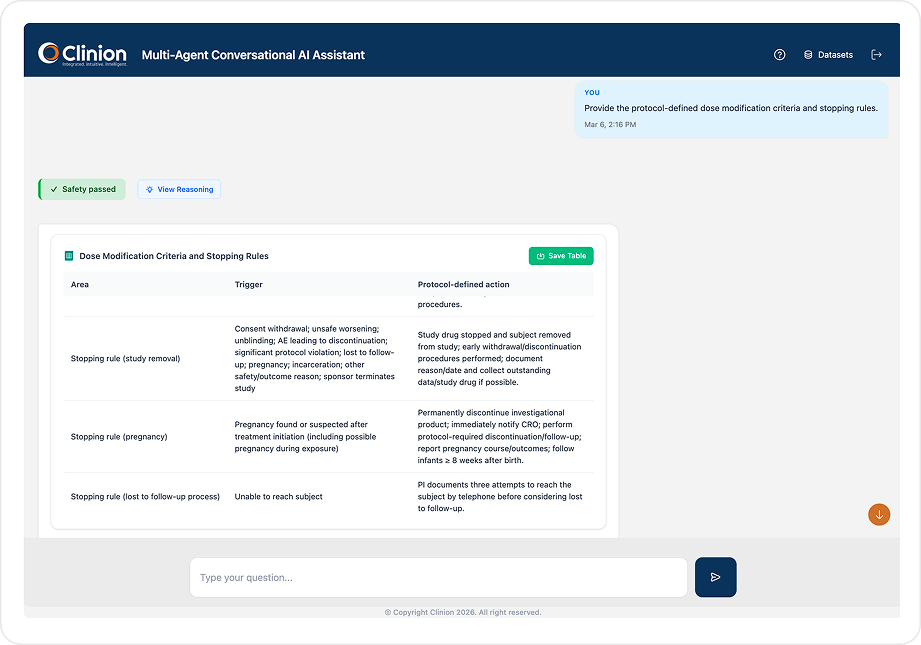
PROTOCOL INTELLIGENCE
Instant Protocol Answers. Grounded. Traceable.
Ask any protocol question in plain English. Agentic Assistant retrieves precise, citation-backed answers from your study-specific knowledge base with full traceability to the original protocol section.
Built automatically when your protocol is uploaded. Indexes eligibility criteria, endpoints, visit schedules, procedures, prohibited medications, SAE timelines, and amendment histories. Every response grounded in validated study documents.
Summarize the efficacy endpoints and list all assessment timepoints with methods.Extract prohibited medications and check if any enrolled subjects have taken them.
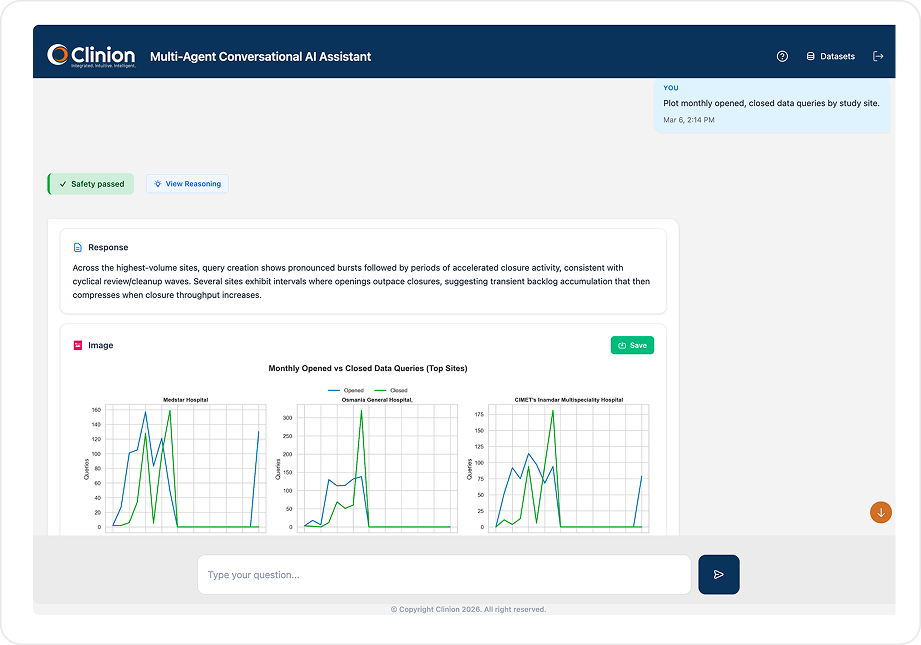
OPERATIONS INSIGHTS
Real-Time Study Metrics. Zero Report Requests.
Ask any operational question and get dynamically calculated metrics from your live EDC – formatted as summaries, tables, or trend comparisons. No reports to pull. No dashboards to configure.
Connects to an operational metadata RAG containing enrollment structures, visit schedules, deviation categories, site performance parameters, and operational KPIs - dynamically calculating metrics like enrollment velocity, visit completion rates, query aging, site-level comparisons, and data entry lag from the live EDC data. Ask follow-up questions to drill deeper into any metric.
Show site-wise enrollment, screen failure rate, and dropout trends for the last 8 weeks. Identify sites with more than 3 protocol deviations this month and categorize the deviation types.
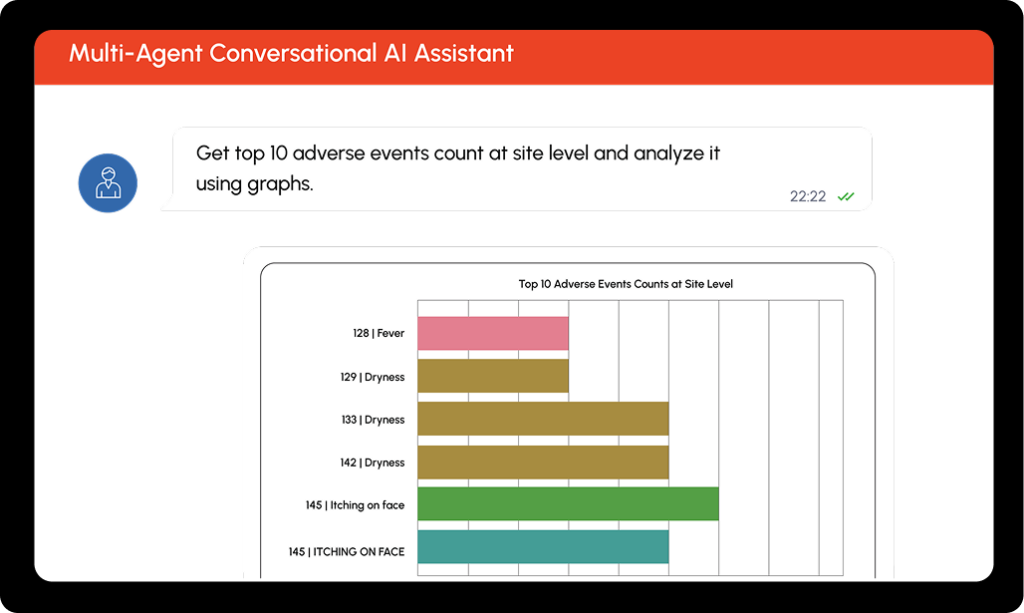
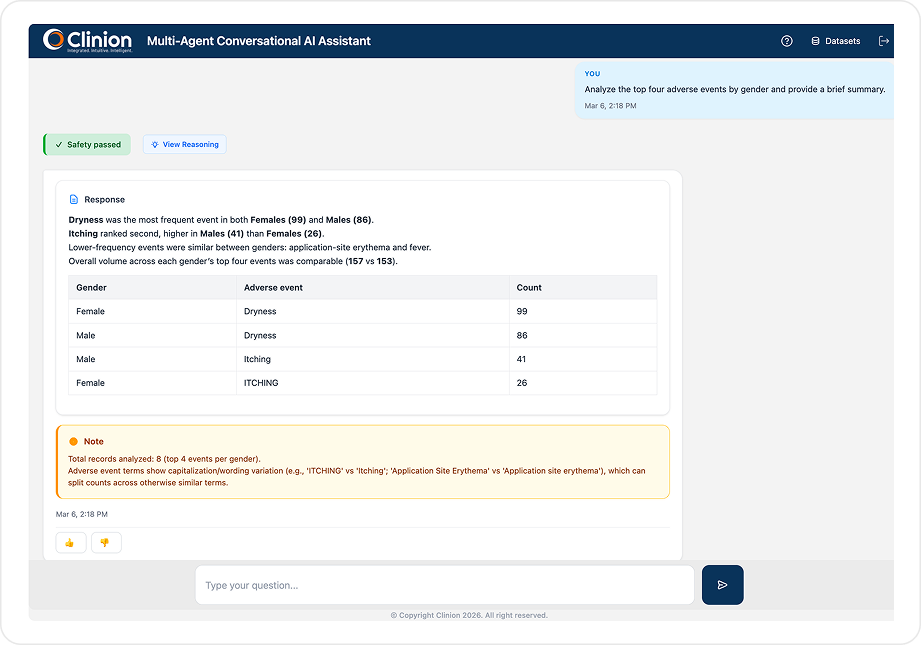
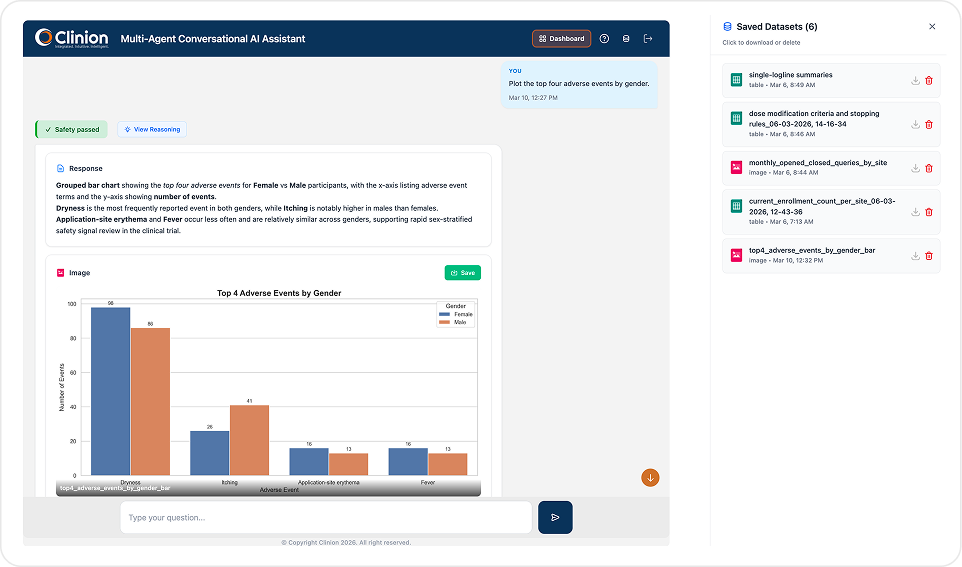
CLINICAL DATA QUERYING
SQL-Free Data Access for Your Entire Study Team
Type what you need in natural language and get formatted, exportable results in seconds. No SQL. No database training. No waiting on someone else to pull the data for you.
Uses an EDC metadata RAG that includes CRF structures, field definitions, visit/variable mappings, controlled terminology, and validation rules to convert natural language prompts into validated queries and retrieve live study data. Returns formatted tables, counts, and summaries - exportable to Excel with one click or saveable as images for reports and presentations.
Fetch subjects with ALT/AST > 3× ULN who also reported adverse events within the same visit window.
How many subjects reported a Grade 3 or higher AE at Visit 6?
EXPORTABLE REPORTS
From Question to Shareable Insight in Minutes
Agentic Assistant doesn’t just answer your question, it packages the answer so you can act on it. Export any data table to Excel for further analysis. Save a response as an image for your next team meeting or monitoring report. Build a library of saved queries your team can rerun as the study progresses. Every output is audit-traceable – linked to the user, the timestamp, and the underlying data source.
ENTERPRISE COMPLIANCE
AI You Can Trust - Controlled, Isolated, Audit-Ready
Deploying AI in clinical trials demands a different standard. Agentic Assistant is built for regulated environments from the ground up. Every query runs within study-level isolation — Study A has zero access to Study B’s data, documents, or configurations. Role-based access controls ensure users only see what they’re authorized to see. All interactions are logged with complete audit traill – aligned with 21 CFR Part 11, EU Annex 11, HIPAA, and GDPR.

Interactive Demo
Explore Agentic Assistant
See how clinical teams use Agentic Assistant to answer protocol questions, surface real-time operational metrics, and query live study data – all through a single conversational interface built directly into the Clinion EDC.
Benefits
Why Clinical Teams Choose Agentic Assistant
80% Faster Data Retrieval
Data retrieval and report generation time reduced by over 80%, enabling teams to focus on analysis and oversight rather than manual data searches across disconnected systems.
Natural Language Data Access
Non-technical users can query the EDC to generate complex datasets directly using simple natural language, removing dependencies on data managers, programmers, or SQL experts.
Cross-Functional Collaboration
Clinical operations, data management, and medical monitoring teams use the same interface to access validated study information ensuring consistency and eliminating version conflicts across departments.
Built-In Regulatory Readiness
All responses are drawn from validated data sources with complete audit traceability, ensuring alignment with GxP and 21 CFR Part 11 principles. No retrofitting compliance after the fact.
Instant Protocol Query Resolution
Protocol questions that used to take 15–20 minutes of manual searching through documents now get answered in seconds. Agentic Assistant’s study-specific RAG delivers precise, citation-backed responses – so teams spend time acting on answers, not looking for them.
Scalable, Modular AI Architecture
The agentic design is built to grow. The modular framework enables future expansion into predictive analytics, automated data quality checks, safety monitoring, and intelligent risk-based monitoring.
FAQS
Frequently Asked Questions (FAQ)
Agentic Assistant is a multi-agent AI assistant built into the Clinion EDC platform. It uses multiple AI agents like Protocol Intelligence, Operations Insights, Clinical Data etc; to answer questions about your study using natural language. Think of it as a knowledgeable colleague who has instant access to your protocol documents, operational metrics, and clinical data, and can give you precise answers in seconds.
Agentic Assistant uses a Retrieval-Augmented Generation (RAG) architecture. Instead of generating answers from general knowledge, it retrieves information directly from your study-specific knowledge base - which is automatically built from your uploaded protocol documents, study configurations, and live EDC data. Every protocol-related answer includes traceable source references so you can verify the response against the original document. For clinical data queries, Agentic Assistant pulls directly from the live EDC database, ensuring the numbers you see are the numbers in your system.
No. Agentic Assistant is designed for clinical professionals, not database engineers. You type your question in plain English the same way you'd ask a colleague, and Agentic Assistant handles the rest. It translates your natural language query into the right data retrieval, whether that's searching a protocol document, calculating an operational metric, or pulling records from the EDC. No SQL. No query syntax. No training required.
Most AI tools in clinical trials address one domain - some focus on data analytics, others on task automation, and others on workflow agents. Agentic Assistant is the only AI assistant that combines protocol intelligence, operational insights, and live clinical data querying in a single conversational interface. More importantly, it's natively built into the Clinion EDC - not a middleware layer or bolt-on tool. This means real-time data access with zero sync delays, and auto-generated knowledge bases that require no setup or manual training.
Yes. Agentic Assistant is built for regulated environments. It features study-level data isolation (Agentic Assistant for Study A cannot access Study B's data), role-based access controls, and complete audit trails for every interaction. It is aligned with 21 CFR Part 11, EU Annex 11, HIPAA, and GDPR requirements. All data processing stays within your study's security perimeter.
Zero setup time. When a new study is created in the Clinion platform, Agentic Assistant automatically generates a study-specific knowledge base by indexing your protocol documents, study configurations, visit schedules, and form structures. There is no manual training, no prompt engineering, and no configuration required. Your AI assistant is ready the moment your study is.
Still have questions?
Explore how Clinion AI can accelerate your trial – reach out to our team.
Responsible AI
Clinion follows Responsible AI principles, ensuring its AI tools are built for safety and reliability, and remains committed to Data Privacy and Security at every step.
- Accountability
- Transparency
- Privacy & Security
- Reliability & Safety
- Fairness
Stop Searching. Start Asking.
Get answers in seconds, not hours. See what Agentic Assistant can do with your study data.
80% faster data retrieval • Seconds per protocol query • Zero SQL required
Compliance
Fully Compliant with Global Standards



