Insights / Blog / Clinical Trials and AI
Beyond Automation: How AI Is Redefining Clinical Data Management
- Abriti Rai
- March 31, 2026

On this Page
- Summary
- The Evolution of Clinical Data Management
- Managing Distributed Clinical Trial Data: Challenges and Modern Solutions
- Why Rule-Based Automation Is No Longer Enough for Clinical Data Management
- How AI Is Changing Clinical Data Review and Pattern Recognition
- AI Applications Across the Clinical Data Management Lifecycle
- Moving Beyond Milestone-Based Data Cleaning: AI-Driven Continuous Data Monitoring
- AI Governance in Clinical Data Management:
- How to Prepare Your CDM Environment for AI Integration
- Measuring the Real Impact of AI in Clinical Data Management
- How AI Is Redefining the Role of Clinical Data Management Professionals
- The Future of AI in Clinical Data Management
- External References
- Summary
- The Evolution of Clinical Data Management
- Managing Distributed Clinical Trial Data: Challenges and Modern Solutions
- Why Rule-Based Automation Is No Longer Enough for Clinical Data Management
- How AI Is Changing Clinical Data Review and Pattern Recognition
- AI Applications Across the Clinical Data Management Lifecycle
- Moving Beyond Milestone-Based Data Cleaning: AI-Driven Continuous Data Monitoring
- AI Governance in Clinical Data Management:
- How to Prepare Your CDM Environment for AI Integration
- Measuring the Real Impact of AI in Clinical Data Management
- How AI Is Redefining the Role of Clinical Data Management Professionals
- The Future of AI in Clinical Data Management
- External References
Summary
Clinical data management is evolving rapidly, and AI is at the center of that change. From continuous data monitoring to smarter anomaly detection, AI is helping CDM teams manage growing complexity across the clinical trial lifecycle while maintaining regulatory compliance and human oversight.
The Evolution of Clinical Data Management
Clinical data management has always adapted to new tools, new regulations, and new study designs. What is different today is not simply the arrival of another technology layer. It is the expectation that data should be usable, reviewable, and interpretable while a study is still underway.
Study teams no longer wait for large data consolidation moments to understand how things are progressing. Decisions about safety, protocol conduct, and operational direction increasingly rely on visibility that is continuous rather than episodic.
This shift has introduced new expectations:
- Insight is needed during the study, not after it.
- Data review must keep pace with incoming information.
- Operational decisions depend on near real-time understanding.
- Workflows designed for sequential review must now function dynamically.
Artificial intelligence has entered this space not as a replacement for established processes, but as a response to how data now behaves. Its role is emerging where manual oversight and rule-based automation alone struggle to maintain pace with incoming information.
Industry analyses project that AI-related spending across healthcare and life sciences could approach 194 billion USD by 2030, with applications tied to clinical development growing at an estimated annual rate above 20 percent.
Managing Distributed Clinical Trial Data: Challenges and Modern Solutions
A decade ago, the majority of trial data moved through tightly controlled capture environments. Today, data arrives from a far broader range of sources, including:
- Electronic health records (EHRs)
- Laboratory systems
- Imaging platforms
- Wearables and connected devices
- Patient-reported channels
- External registries and datasets
Observations suggest that only 30 to 60 percent of study data now originates directly within traditional capture systems. The remainder must be integrated, standardized, and contextualized before it becomes analysis-ready.
Operational Impact of Distributed Data
As data sources expand, operational models must shift from linear handling to coordinated data orchestration.
Traditional Model | Current Environment |
Data primarily entered into one system | Data originates across multiple ecosystems |
Structured inputs dominate | Structured and unstructured inputs coexist |
Review follows data entry | Review requires alignment before evaluation |
Reconciliation is limited | Continuous reconciliation becomes necessary |
These realities reflect how research itself has evolved. Studies are drawing on richer evidence streams, and workflows must adapt accordingly.
Why Rule-Based Automation Is No Longer Enough for Clinical Data Management
Automation has already improved clinical data management in meaningful ways:
- Edit checks reduced repetitive validation.
- Data transfer routines accelerated aggregation.
- Standard workflows improved consistency.
Yet most automation relies on predefined rules. It executes what it has been instructed to look for. It does not evaluate context, identify emerging relationships, or adapt when patterns shift.
As data diversity increased, teams encountered limits:
- Queries still required manual interpretation.
- Cross-dataset comparisons demanded human judgment.
- Pattern recognition depended heavily on individual expertise.
- Automation accelerated execution but did not change how insight was discovered.
This distinction explains why organizations began exploring AI capabilities that could support interpretation alongside execution.
How AI Is Changing Clinical Data Review and Pattern Recognition
AI enters clinical data management most effectively when applied to pattern recognition and contextual evaluation rather than task replacement.
Examples of Workflow-Level Change
- Machine learning models examine datasets across sites to identify subtle deviations.
- Natural language processing assists with interpreting unstructured inputs.
- Context-aware systems support more consistent query generation.
- Continuous comparison across datasets replaces periodic validation cycles.
Across implementations, organizations have reported measurable reductions in manual error-checking workloads. The gain is not simply speed. It is the ability to redirect expertise toward evaluation rather than detection.
AI Applications Across the Clinical Data Management Lifecycle
AI applications are already shaping how data teams manage complexity, helping translate fragmented inputs into actionable information while keeping expert review firmly in place.
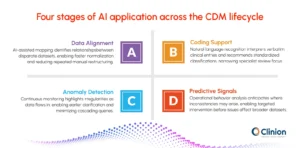
Data Alignment
With information arriving from labs, devices, imaging platforms, and external databases, aligning formats can be time-intensive. AI-assisted mapping identifies relationships between disparate datasets, enabling faster normalization and reducing the need for repeated manual restructuring.
Coding Support
Natural language recognition can interpret verbatim clinical entries and recommend standardized classifications. This streamlines the coding workflow by narrowing the review focus, allowing specialists to validate rather than search from scratch.
Anomaly Detection
Continuous pattern analysis highlights irregularities as data flows in, bringing attention to deviations that might otherwise remain hidden until later review stages. Earlier awareness supports quicker clarification and minimizes cascading queries.
Predictive Signals
Trend-based models assess operational behaviors to anticipate where inconsistencies or quality concerns may arise. This foresight enables targeted follow-up, helping teams intervene before issues affect broader datasets.
Collectively, these capabilities allow data managers to stay ahead of volume and variability, reinforcing accuracy while preserving the critical role of human judgment
Agentic AI in Clinical Trials: Orchestrating Data Workflows at Scale
A further evolution now being explored is the use of agentic AI models designed to manage defined operational sequences rather than perform isolated analyses. These systems do not make autonomous clinical decisions. Instead, they can initiate configured actions such as assembling data views, triggering reconciliation workflows, or flagging downstream dependencies when new information enters the environment.
In this model, AI acts less like a tool and more like an orchestrator operating within strict boundaries. Each action remains rules-governed, auditable, and reviewable by data management teams. The objective is not automation of judgment, but reduction of the coordination burden created by increasingly interconnected data streams.
Early exploration suggests that such approaches may help organizations manage workflow continuity across platforms while maintaining the same validation and oversight structures already required in regulated settings.
Moving Beyond Milestone-Based Data Cleaning: AI-Driven Continuous Data Monitoring
Traditional workflows concentrated effort around defined milestones such as interim analyses or database lock preparation. AI-supported environments encourage a different rhythm.
Milestone-Centric Approach | Continuous Awareness Model |
Data cleaned in phases | Data evaluated continuously |
Issues accumulate before resolution | Signals addressed earlier |
Intensive late-stage review | Distributed oversight |
Database lock requires concentrated effort | Database readiness becomes progressive |
This approach reshapes timelines without eliminating milestones. It creates a smoother path toward them.
AI Governance in Clinical Data Management:
Introducing AI into regulated research settings calls for disciplined governance that matches the sensitivity of clinical data. The priority is not just performance, but demonstrable control over how insights are generated, reviewed, and documented.
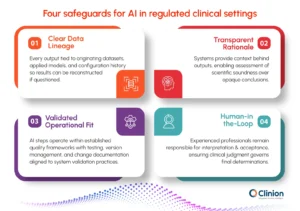
Organizations adopting AI should embed safeguards that ensure:
Clear data lineage
Every output must be tied back to its originating datasets, applied models, and configuration history so results can be reconstructed if questioned.
Transparent rationale
Systems should present the context behind recommendations, enabling reviewers to assess relevance and scientific soundness rather than relying on opaque conclusions.
Validated operational fit
AI-enabled steps need to operate within established quality frameworks, including testing, version management, and change documentation aligned with existing system validation practices.
Qualified human adjudication (Human-in-the-loop)
Experienced professionals remain responsible for interpretation and acceptance, ensuring that clinical judgment governs final determinations.
Regulatory guidance reinforces this structure. Bodies such as the U.S. Food and Drug Administration and the International Council for Harmonisation continue to stress verifiable processes, data reliability, and accountability independent of the technologies employed.
In this context, AI serves as an analytical augmentation. It helps organize complexity and surface insights efficiently, while established oversight mechanisms preserve responsibility, audit readiness, and confidence in the evidence generated.
How to Prepare Your CDM Environment for AI Integration
Organizations typically begin by assessing:
Processes with heavy manual reconciliation
Activities that require frequent cross-checking across datasets often consume disproportionate time. These are strong candidates for AI-assisted comparison and pattern matching, allowing teams to focus on resolving exceptions instead of locating them.
Workflows involving repetitive interpretation
Tasks such as reviewing similar data patterns, classifying entries, or applying consistent rules across large volumes can benefit from models trained to recognize context, reducing review fatigue while maintaining consistency.
Compatibility with existing platforms such as EDC and safety systems
AI initiatives must align with the current technology landscape. Integrations should extend system value rather than introduce parallel workflows, ensuring continuity in data flow, audit trails, and user experience.
Governance structures for validation and monitoring
Clear procedures are needed to define how models are evaluated, updated, and overseen. This includes performance checks, documentation standards, and defined ownership for ongoing supervision.
Preparing teams to assess AI-assisted outputs responsibly
Training shifts from performing tasks manually to critically reviewing machine-supported insights. Data professionals must understand system behavior well enough to validate results and recognize when further scrutiny is required.
Successful adoption is therefore as much an operational discipline as a technical deployment. The goal is to embed AI where it alleviates friction, strengthens quality oversight, and complements the expertise already present within CDM teams.
Measuring the Real Impact of AI in Clinical Data Management
While time savings are valuable, many organizations observe that the more meaningful impact lies in stability.
Observed Operational Improvements
- Reduced buildup of unresolved discrepancies, preventing last-minute escalations.
- Reconciliation activities becoming more routine and less disruptive.
- Earlier visibility into study status, enabling timely coordination across functions.
- Greater predictability when preparing for analysis and submission readiness.
These changes influence the rhythm of study execution. Even when not reflected in a single performance metric, they contribute to a more consistent and less reactive data management environment.
How AI Is Redefining the Role of Clinical Data Management Professionals
As AI assumes greater responsibility for identifying patterns and surfacing signals, the role of clinical data managers is evolving toward higher-level evaluation.
Responsibilities Are Expanding Toward:
- Interpreting surfaced signals within clinical and operational context.
- Safeguarding consistency across increasingly connected datasets.
- Engaging earlier with clinical and statistical stakeholders to align expectations.
- Providing oversight to data that is reviewed continuously rather than in stages.
This progression enhances the role rather than reducing it. Expertise, judgment, and regulatory awareness remain central, while AI broadens the capacity of teams to manage scale without diluting accountability.
The Future of AI in Clinical Data Management
Clinical data management continues to adapt to the realities of modern research, where information flows continuously and expectations for visibility remain high. Artificial intelligence is becoming integrated into this environment as a means of supporting interpretation, alignment, and sustained oversight.
Organizations are refining how established principles are carried out in a landscape shaped by broader data sources and faster decision cycles. As adoption progresses, AI will likely become less of a distinct topic and more of an embedded component of everyday data practice.
Understanding Clinical Data Management Challenges
Explore the key challenges impacting clinical data management today and how they influence trial efficiency, data quality, and compliance.
Read the full article →
External References

Abriti Rai writes on the intersection of AI, automation, and clinical research. At Clinion, she develops content that simplifies complex innovations and highlights how technology is shaping the next generation of data-driven clinical trials.
FAQS
Frequently Asked Questions
AI in clinical data management uses machine learning (ML) and natural language processing (NLP) to support data review, anomaly detection, and workflow coordination in clinical trials. It surfaces insights efficiently while keeping qualified professionals in control of final decisions.
AI continuously monitors incoming data to detect inconsistencies and anomalies earlier than traditional review cycles would allow. This helps teams resolve issues faster and maintain dataset reliability throughout the study.
Common applications include automated data alignment, AI-assisted medical coding, real-time anomaly detection, and predictive trend analysis. Each reduces manual workload while keeping expert review firmly in place.
Yes, when implemented with clear data lineage, transparent model rationale, and qualified human oversight at every decision point. Regulatory bodies like the FDA and ICH continue to emphasize accountability regardless of the technology used.
AI enables ongoing data evaluation as information arrives rather than concentrating review around study milestones. This reduces late-stage data cleaning and minimizes last-minute escalations before submission.
Agentic AI manages defined operational task sequences such as triggering reconciliation workflows or flagging dependencies when new data arrives. All actions remain auditable and governed by rules set by the data management team.
Organizations should identify workflows with heavy manual reconciliation and assess compatibility with existing platforms like EDC and safety systems. Teams also need training to critically evaluate AI-assisted outputs rather than accepting them without scrutiny.
As AI handles more pattern detection, clinical data managers are shifting toward higher-level evaluation and strategic oversight responsibilities. The role is expanding in influence rather than being reduced by automation.
Still have questions?
Explore how Clinion AI can accelerate your trial – reach out to our team.
Unlock the Future of Clinical Trials with Clinion.
Cut your trial costs by 35% and accelerate your time-to-market by 30%
Compliance
Fully Compliant with Global Standards


