Insights / Blog / EDC
Clinion EDC vs Medidata Rave: Evaluating EDC Platforms for Modern Clinical Trials
- Abriti Rai
- March 20, 2026

On this Page
- Summary
- The Expanding Role of EDC Platforms
- Overview of Medidata Rave
- Overview of Clinion EDC
- Evaluating the EDC Platforms
- Platform Architecture and Integrations
- When Should Sponsors Evaluate Alternative EDC Platforms?
- Final Considerations
- External Reference
- Summary
- The Expanding Role of EDC Platforms
- Overview of Medidata Rave
- Overview of Clinion EDC
- Evaluating the EDC Platforms
- Platform Architecture and Integrations
- When Should Sponsors Evaluate Alternative EDC Platforms?
- Final Considerations
- External Reference
Summary
Electronic Data Capture platforms determine how efficiently clinical trial data is collected, reviewed, and prepared for regulatory submission. Every patient record, adverse event, laboratory value, and protocol-defined variable eventually enters the EDC system. Because of this, the choice of platform directly affects study timelines, operational effort, and data quality.
Clinical research, however, has evolved significantly. Trials now involve decentralized data sources, wearable devices, remote monitoring, and increasingly large datasets. These changes have prompted sponsors and CROs to evaluate newer platforms that offer stronger automation, faster study configuration, and more integrated workflows.
As a result, the market now includes a wide range of EDC solutions designed to support modern trial requirements. This article compares Medidata Rave and Clinion EDC across key areas, including study setup, data review, reporting, integrations, and operational efficiency.
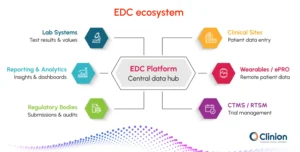
The Expanding Role of EDC Platforms
Early EDC systems primarily replaced paper case report forms with digital equivalents. This change improved accessibility and reduced transcription errors.
Over time, additional capabilities were introduced, including:
- edit checks and validation rules
- query generation and resolution
- audit trails and compliance monitoring
- reporting and analytics tools
Clinical trials now require EDC systems to support more complex workflows. Modern platforms are expected to handle multiple data sources, integrate with other clinical systems, and provide faster access to study insights.
These expectations have increased interest in platforms that automate repetitive tasks and reduce dependence on manual processes.
Overview of Medidata Rave
Medidata Rave EDC is one of the most widely adopted electronic data capture systems in the clinical research industry. It has been used in thousands of studies across therapeutic areas and geographic regions.
Core capabilities include:
- electronic case report form design
- data entry validation and discrepancy checks
- query management workflows
- audit trails for regulatory compliance
- reporting dashboards and analytics
Rave also integrates with other solutions within the Medidata ecosystem, enabling organizations to manage several aspects of clinical development within a connected system environment.
Because many research sites have prior experience with the platform, training requirements may be lower for organizations that continue using it.
At the same time, certain configuration tasks often require specialized programming expertise, which can affect setup timelines and operational flexibility.
Overview of Clinion EDC
Clinion EDC represents a newer generation of EDC platforms designed to support automation and integrated clinical workflows.
The system applies artificial intelligence (AI) across several stages of clinical data management. Instead of focusing only on data capture, it supports processes such as study configuration, discrepancy detection, reporting, and coding.
Key capabilities include:
- AI-assisted study configuration
- automated CDASH variable mapping
- discrepancy identification during data review
- automated reporting through natural language prompts
- conversational assistance for platform users
Clinion operates within a unified eClinical environment that connects EDC with modules such as randomization, ePRO, CTMS, eConsent, and eSource. This structure allows information to move across systems without extensive reconciliation processes.
Evaluating the EDC Platforms
To understand how these systems support modern trials, it is important to examine their capabilities in detail. The following sections compare Medidata Rave EDC and Clinion EDC across the operational areas that influence study execution.
Feature Comparison
Capability | Clinion EDC | Medidata Rave |
|---|---|---|
Study Configuration | Codeless with AI/CDASH libraries; fast deployment | Templates; often needs programmers |
Data Review | Agentic AI detects discrepancies; 90% less effort | Rule-based checks; manual query cycles |
Protocol Amendments | Seamless updates, version control | Supported with technical coordination |
Reporting | Natural language AI generation | Dashboards, custom exports |
AI Integration | Broad: agents, mapping, coding, conversational | Targeted: ML coding, study builds |
Architecture | Unified eClinical (RTSM, CTMS, etc.) | Medidata ecosystem modules |
Study Setup and Configuration
Database configuration is often one of the longest stages during study startup. Teams must design case report forms, configure validation rules, and align variables with CDISC standards.
Medidata Rave
Rave supports study configuration through reusable templates and predefined objects. Teams can reuse forms and logic from previous studies to reduce setup effort.
However, configuring complex validation rules and workflows may require database programmers or vendor support. For large or complex trials, coordination between multiple teams can extend configuration timelines.
Clinion EDC
Clinion uses standardized global libraries and codeless configuration tools to simplify study setup.
Users can configure case report forms, validation rules, and workflows directly within the platform. Prebuilt templates aligned with CDASH standards allow teams to assemble study databases more efficiently.
Because configuration does not rely heavily on programming, study teams can manage many tasks independently and deploy studies faster.
Data Review and Discrepancy Management
Data cleaning consumes a large portion of the effort involved in clinical data management. Detecting discrepancies, issuing queries, and coordinating with sites often requires multiple review cycles.
Medidata Rave
Rave uses edit checks and validation rules to detect discrepancies during data entry. When issues are identified, queries are generated and sent to study sites for resolution.
This approach supports structured data review but still depends on repeated communication between sites and data managers.
Clinion EDC
Clinion applies AI to assist with discrepancy detection and dataset generation.
Data managers can create datasets using natural language prompts and identify inconsistencies directly within the system. Queries or corrections can be issued across multiple subjects simultaneously, reducing repetitive actions.
These capabilities can shorten data review cycles and help teams reach database lock more efficiently.
Managing Protocol Amendments
Protocol amendments occur frequently during clinical trials. EDC platforms must support configuration updates without compromising existing data.
Medidata Rave
Rave allows mid-study updates to forms and workflows. Implementing these changes may require coordination with technical teams to ensure proper validation and deployment.
Clinion EDC
Clinion enables study teams to modify visits, pages, and data fields after study launch.
The system maintains version control to protect existing data while applying updates. Configuration changes can be implemented without interrupting ongoing study activity.
Reporting and Data Access
Clinical teams rely on reporting tools to track study progress, review safety signals, and prepare regulatory submissions.
Medidata Rave
Rave includes reporting dashboards that provide visibility into data entry progress, query resolution rates, and site activity.
Custom reporting may require additional configuration depending on organizational workflows.
Clinion EDC
Clinion includes an AI-based reporting engine that generates custom reports from simple prompts.
Users can request reports directly within the platform without writing queries or scripts. Automated alerts and audit trails support regulatory documentation and internal oversight.
Artificial Intelligence and Automation
Automation is becoming an increasingly important capability within clinical trial technology.
Medidata Rave
Medidata has introduced machine learning capabilities in certain modules, including automated medical coding.
These tools can improve efficiency in specific workflows but are typically implemented alongside traditional data management processes.
Clinion EDC
Clinion incorporates AI across multiple workflows, including:
- Discrepancy Detection
AI analyzes incoming study data to identify inconsistencies, missing values, and potential protocol deviations. This allows data managers to detect and address issues earlier in the review process. - CDASH Mapping
AI suggests mappings between study variables and CDASH standards based on field definitions and context. Users can quickly review and apply these mappings, reducing manual effort. - Medical Coding
AI assists in matching reported medical terms with standardized dictionaries such as MedDRA and WHODrug. This improves coding efficiency and consistency. - Report Generation
Natural language prompts are converted into structured reports within the platform.
Users can retrieve study insights without writing queries or scripts. - Conversational User Support
An AI assistant provides contextual guidance and answers user queries directly within the system. This helps users complete tasks without external support.
These capabilities reduce repetitive tasks and support faster decision-making during data review.
Agentic AI in Clinical Data Management
A newer development in clinical trial technology is the use of agentic AI systems. Unlike traditional automation, agentic AI uses intelligent agents that can monitor incoming data, analyze patterns, and recommend actions to users.
Clinion EDC incorporates agentic AI through specialized agents designed to support different aspects of clinical data management.
Agentic AI for Data Review
The Agentic AI Data Review tool reads the study protocol and CRFs to understand study structure and expected data relationships. It continuously analyzes incoming study data and automatically generates prompts and queries when discrepancies are detected. Data managers only review and approve the suggested queries, after which they are created directly in the EDC.
Conversational AI with Multi-Agent Support
The platform also includes a Conversational AI Assistant interface powered by multiple specialized agents. These agents can interpret user questions, analyze study datasets, retrieve relevant information from CRFs, and generate reports or insights. This allows users to interact with study data using natural language instead of navigating multiple system workflows.
Managing Imaging Data and Confidential Information
Clinical trials often involve imaging data that must be handled carefully to protect patient privacy.
Clinion includes a medical image repository with AI-based redaction capabilities. The system automatically detects and removes patient identifiers and confidential information from imaging files. This process can be completed in seconds, allowing imaging data to be stored and shared securely across study teams.
Imaging workflows in other platforms may involve separate modules or additional integrations depending on study requirements.
Platform Architecture and Integrations
Clinical trials rely on several interconnected systems, including randomization platforms, patient reporting tools, and trial management systems.
Medidata Rave
Rave integrates with applications within the Medidata platform environment, allowing sponsors to manage multiple aspects of clinical development.
These modules can operate as connected systems within the broader ecosystem.
Clinion EDC
Clinion uses a unified architecture where modules such as RTSM, CTMS, eConsent, ePRO, eSource, eProtocol Automation, CSR Automation, and eTMF operate within the same platform environment. Because these systems share a common structure, data flows directly between modules without extensive reconciliation or integration work.
When Should Sponsors Evaluate Alternative EDC Platforms?
Organizations often reconsider their EDC systems when operational challenges begin affecting study performance.
Common triggers include:
- long study configuration timelines
- increasing database programming costs
- extended data cleaning cycles
- limited automation capabilities
- multiple disconnected systems requiring reconciliation
When these issues become frequent, evaluating newer platforms may help improve operational efficiency.
Final Considerations
Both Medidata Rave EDC and Clinion EDC provide comprehensive capabilities for managing clinical trial data.
Medidata Rave has a long track record of supporting global studies and remains widely used across the industry.
At the same time, modern trials increasingly require platforms that reduce manual configuration, automate data management processes, and provide faster access to study insights.
Organizations evaluating EDC systems today must consider how effectively a platform supports the entire clinical data management lifecycle, from study setup through database lock and reporting.
External Reference

Abriti Rai writes on the intersection of AI, automation, and clinical research. At Clinion, she develops content that simplifies complex innovations and highlights how technology is shaping the next generation of data-driven clinical trials.
FAQS
Frequently Asked Questions
The key difference lies in how the platforms handle study setup and data workflows.
Medidata Rave follows a traditional model with configured builds and established processes.
Clinion EDC focuses on AI-driven automation, faster setup, and a unified platform with integrated modules.
Yes, both platforms are designed to support regulations such as 21 CFR Part 11. They include audit trails, electronic signatures, and role-based access controls. Compliance depends on proper system validation and study-level implementation.
Ease of use depends on user familiarity and system design. Medidata Rave is widely used, so many users are already trained on it. Clinion EDC emphasizes simplified workflows and codeless configuration to reduce user effort.
Costs vary based on study scope, integrations, and support requirements. Traditional systems may involve additional effort for programming and mid-study changes. Platforms with automation and built-in modules can help reduce overall operational costs.
Study setup in Medidata Rave EDC typically involves programming, validation cycles, and multiple review rounds. This can extend timelines depending on study complexity.
Clinion EDC uses codeless configuration and standardized libraries to enable faster deployment.
Yes, Clinion offers integrated modules such as RTSM, eProtocol Generation, ePRO, CTMS, eConsent, eSource, CSR Automation, and eTMF. These modules operate within a single platform to streamline workflows. This reduces dependency on external integrations and improves data visibility.
Scalability depends on system architecture and the ability to support multi-site studies.
Medidata Rave has a long track record in large global trials. Other platforms are also designed to scale while focusing on efficiency and integration.
The choice depends on how efficiently CROs can manage multiple studies and timelines.
Key factors include study setup speed, flexibility, and ease of handling changes. Platforms with automation and faster configuration may improve operational efficiency.
Clinical trials now require faster execution and reduced manual effort. Organizations are looking for platforms with stronger automation and integrated workflows. This has led to increased evaluation of newer EDC solutions alongside established systems.
Still have questions?
Explore how Clinion AI can accelerate your trial – reach out to our team.
Unlock the Future of Clinical Trials with Clinion.
Cut your trial costs by 35% and accelerate your time-to-market by 30%
Compliance
Fully Compliant with Global Standards


