Insights / Blog / Medical Imaging
Medical Imaging in Clinical Trials: Understanding DICOM and the Role of AI
- Abriti Rai
- March 26, 2026
On this Page
- Summary
- What Is Medical Imaging in Clinical Trials?
- Why Medical Imaging Is Critical in Clinical Trials
- Types of Medical Imaging Used in Clinical Trials
- How Medical Imaging Data Is Used to Measure Treatment Response
- Imaging Biomarkers in Clinical Trials: What They Are and Why They Matter
- Challenges in Medical Imaging for Clinical Trials
- What Is DICOM? An Explanation for Clinical Trials
- Why DICOM Is Important in Clinical Trials
- How DICOM Standardizes Imaging Data Across Clinical Trials
- Key Challenges in Managing DICOM Imaging Data
- Role of AI in Medical Imaging for Clinical Trials
- The Evolving Role of Medical Imaging in Clinical Trials
- External Reference
- Summary
- What Is Medical Imaging in Clinical Trials?
- Why Medical Imaging Is Critical in Clinical Trials
- Types of Medical Imaging Used in Clinical Trials
- How Medical Imaging Data Is Used to Measure Treatment Response
- Imaging Biomarkers in Clinical Trials: What They Are and Why They Matter
- Challenges in Medical Imaging for Clinical Trials
- What Is DICOM? An Explanation for Clinical Trials
- Why DICOM Is Important in Clinical Trials
- How DICOM Standardizes Imaging Data Across Clinical Trials
- Key Challenges in Managing DICOM Imaging Data
- Role of AI in Medical Imaging for Clinical Trials
- The Evolving Role of Medical Imaging in Clinical Trials
- External Reference
Summary
Medical imaging is widely used in clinical trials to assess disease progression and treatment response. However, reliability demands standardization and consistent interpretation across sites. DICOM enables this by standardizing image storage, sharing, and metadata. Meanwhile, artificial intelligence enhances analysis, quality control, and anonymization. Together, they transform captured data into actionable insights for clinical research.
What Is Medical Imaging in Clinical Trials?
Medical imaging in clinical trials refers to the use of imaging modalities such as CT, MRI, PET, and ultrasound to generate visual and quantitative data that support research objectives. Unlike routine clinical imaging, which focuses on diagnosing and managing individual patients, imaging in clinical trials is designed to generate standardized, comparable data across multiple participants and sites.
In a clinical trial setting, imaging is not just about viewing anatomy. It is used to capture quantifiable evidence that can be analyzed across time points, treatment groups, and geographic regions.
Key characteristics of imaging in clinical trials:
- Standardized imaging protocols across all sites
- Repeatable measurements across multiple visits
- Direct alignment with study endpoints and objectives
- Structured data collection to support regulatory review
Why Medical Imaging Is Critical in Clinical Trials
Imaging plays a central role in how clinical trials are designed and evaluated. It supports decision-making at every stage, from selecting the right participants to determining whether a treatment is effective.
Role of Imaging Across Trial Stages
Trial Stage | Role of Imaging | Impact |
Pre-enrollment | Assess eligibility and confirm disease presence | Improves patient selection |
Baseline | Establish initial disease status | Enables accurate comparison |
During trial | Monitor disease progression or response | Detects early treatment signals |
Endpoint | Evaluate outcomes | Supports objective conclusions |
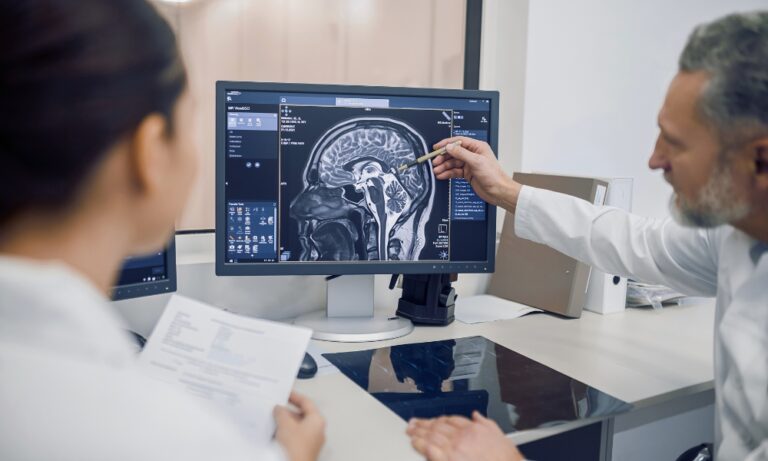
Types of Medical Imaging Used in Clinical Trials
Different imaging modalities are used in clinical trials depending on what needs to be measured, each technique providing a distinct type of insight.
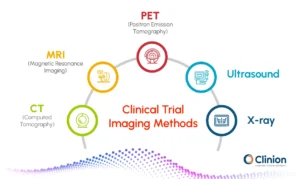
Common imaging modalities used in clinical trials:
CT (Computed Tomography):
CT scans are commonly used in clinical trials, especially in oncology, to measure tumor size and monitor changes over time. They provide detailed cross-sectional images of the body, making them reliable for tracking disease progression and evaluating treatment response using standardized criteria.
MRI (Magnetic Resonance Imaging):
MRI is used for detailed imaging of soft tissues such as the brain, muscles, and organs. It is particularly valuable in neurological and musculoskeletal studies, where high contrast between different tissue types is required. MRI can also support advanced techniques that provide insights beyond basic structure.
PET (Positron Emission Tomography):
PET scans are used to assess metabolic and functional activity within tissues. Instead of just showing structure, PET helps identify how active a disease is at a cellular level, which can be useful for detecting early treatment response even before physical changes are visible.
Ultrasound:
Ultrasound uses sound waves to produce real-time images and is often used for monitoring organs, blood flow, or guiding procedures. It is non-invasive, widely accessible, and does not involve radiation, making it suitable for repeated use in certain studies.
X-ray:
X-rays are primarily used for structural assessment, particularly in bone-related conditions and chest imaging. While less detailed than other modalities, they remain useful for specific trial requirements where quick and simple imaging is sufficient.
In many clinical trials, multiple imaging modalities are used together to capture both structural and functional changes, providing a more complete and reliable understanding of disease progression and treatment impact.
How Medical Imaging Data Is Used to Measure Treatment Response
In clinical trials, imaging is used to track how a disease changes over time and whether a treatment is having the intended effect. Instead of relying only on symptoms or lab values, imaging provides a direct way to observe and measure these changes.
For example, tumor size can be tracked across visits, or metabolic activity can be assessed to understand how a disease responds to treatment. These measurements form the basis for evaluating treatment effectiveness.
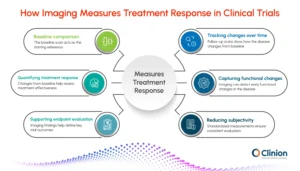
How imaging data is used in practice:
Establishing a baseline:
The first set of images captured at the start of the trial defines the initial disease burden. This serves as the reference point against which all future changes are compared.
Tracking changes across visits:
Follow-up scans are performed at predefined intervals to monitor how the disease evolves. Measurements such as tumor size, lesion count, or tissue characteristics are compared with the baseline to identify trends.
Quantifying treatment response:
Imaging allows researchers to classify responses based on measurable changes, such as a reduction in tumor size, no significant change, or disease progression. This helps determine whether the treatment is effective.
Capturing functional and biological changes:
In some cases, imaging goes beyond structure and captures functional activity, such as metabolism or blood flow. These changes can indicate a response earlier than visible structural changes.
Supporting endpoint evaluation:
Imaging findings are used to define key trial outcomes, such as response rate or progression. These outcomes are critical for evaluating the success of the treatment.
Reducing subjectivity in assessments:
By relying on measurable criteria, imaging helps standardize how the response is evaluated across different investigators and sites.
Standardized response frameworks are then applied to ensure that these assessments remain consistent across all participants and sites.
Imaging Biomarkers in Clinical Trials: What They Are and Why They Matter
maging biomarkers are quantifiable features extracted from medical images that are used to assess disease characteristics or treatment response. Instead of relying only on visual interpretation, these biomarkers turn images into measurable data points that can be tracked over time.
In clinical trials, imaging biomarkers are often derived by comparing scans against a baseline, allowing researchers to detect even small changes that may not be immediately visible.
Where imaging biomarkers are used:
- Tumor measurement:
Changes in tumor size or volume are used to evaluate whether a treatment is working. - Lesion characterization:
Features such as shape, density, or texture help differentiate between disease types or stages. - Functional activity assessment:
In modalities like PET, biomarkers reflect metabolic activity, offering early signals of response. - Longitudinal tracking:
Biomarkers are measured across timepoints to monitor progression or stability relative to baseline.
Why imaging biomarkers matter in trials:
- Enable earlier insights: Detect treatment response before clinical symptoms change
- Support data-driven decisions: Provide measurable evidence rather than visual judgment alone
- Improve consistency: Standardized measurements reduce variability across sites
- Strengthen endpoints: In some studies, biomarkers are used to support or define trial outcomes
In certain cases, validated imaging biomarkers can act as surrogate endpoints, helping accelerate study timelines while maintaining scientific rigor.
Challenges in Medical Imaging for Clinical Trials
Despite its value, medical imaging in clinical trials comes with operational and data-related challenges that need to be managed carefully.
Challenge | What It Means in Practice |
Variability across sites and devices | Different scanners and equipment can affect image quality and consistency |
Non-uniform imaging protocols | Differences in how scans are performed make comparisons difficult |
Reader variability | Interpretations may vary between radiologists, impacting consistency |
High data volume | Imaging generates large, complex datasets that require structured management |
Patient privacy concerns | Imaging data may contain identifiable information, requiring secure handling and redaction |
These challenges make it critical to have standardized processes and robust systems in place to ensure that imaging data remains consistent, secure, and usable across the trial.
What Is DICOM? An Explanation for Clinical Trials
DICOM (Digital Imaging and Communications in Medicine) is the global standard used to manage medical imaging data across systems and sites.
Before understanding DICOM, it is important to make one distinction clear.
Imaging technologies such as CT, MRI, and PET are used to generate medical images.
DICOM defines how those images are stored, structured, and shared.
A DICOM file contains both:
- The image itself
- Metadata that provides context
This metadata includes details such as patient information, scan parameters, device settings, and study identifiers.
What makes DICOM essential:
- Standardized format for all imaging data
- Embedded metadata for context and traceability
- Seamless data exchange across systems
- Support for regulatory compliance
Why DICOM Is Important in Clinical Trials
Clinical trials often involve multiple sites, imaging systems, and device manufacturers. Without a common standard, ensuring that imaging data remains consistent and usable across all locations becomes extremely difficult.
Without vs With DICOM in Clinical Trials
Without DICOM | With DICOM |
Inconsistent image formats across sites | Standardized format used across all systems |
Limited interoperability between systems | Seamless data exchange across platforms and vendors |
Missing or incomplete contextual data | Complete metadata, including patient, study, and acquisition details |
Manual effort to interpret and organize data | Structured data that is ready for analysis and integration |
Higher risk of errors and misinterpretation | Improved accuracy and consistency in interpretation |
How DICOM Standardizes Imaging Data Across Clinical Trials
DICOM enables standardization at multiple levels, in order to ensure:

- Uniform image formatting across all sites:
All images follow the same structure, regardless of the scanner or manufacturer, making them easier to store, view, and analyze. - Consistent capture of imaging parameters:
Details such as slice thickness, acquisition settings, and modality-specific parameters are recorded in a standardized way, enabling accurate comparisons. - Reliable data transfer between systems:
DICOM allows seamless exchange of imaging data across platforms, reducing the need for manual intervention or format conversion. - Preservation of both image and context:
Each image is stored along with rich metadata, including patient details, study information, and acquisition context, ensuring nothing is lost during analysis.
This level of standardization makes it possible to confidently analyze imaging data across large, multi-center trials, where consistency and data integrity are critical.
Key Challenges in Managing DICOM Imaging Data
Even with DICOM in place, managing imaging data in clinical trials remains complex due to the scale, sensitivity, and structure of the data involved.
Key challenges include:
Large file sizes:
High-resolution imaging files require significant storage capacity and can slow down data transfer across sites.
Complex metadata management:
DICOM files include extensive metadata that must be accurately handled to ensure correct interpretation and traceability.
Patient data anonymization:
Identifiable information embedded within images and metadata must be carefully removed or masked to maintain compliance.
Manual and fragmented workflows:
Tasks such as data validation, organization, and quality checks often require manual effort, increasing the risk of delays and errors.
These challenges highlight the need for more advanced, automated solutions that can streamline imaging workflows while maintaining data integrity and compliance.
Role of AI in Medical Imaging for Clinical Trials
Artificial intelligence is increasingly being used to improve how imaging data is managed in clinical trials.
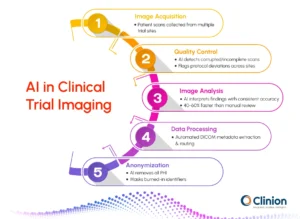
AI in Image Analysis
AI enables faster and more consistent interpretation of imaging data by analyzing large volumes of scans with minimal variability. It supports:
- Detection of subtle patterns that may not be easily visible
- Consistent interpretation across readers and sites
- Faster turnaround times for image review and assessment
AI in Data Processing and Workflow Automation
Beyond analysis, AI plays a key role in streamlining how imaging data is handled operationally. It enables:
- Automated extraction and structuring of DICOM metadata
- Streamlined image ingestion, organization, and routing
- Reduced reliance on manual workflows and data handling
AI in DICOM Redaction and Anonymization
DICOM files often contain sensitive patient information, both within metadata fields and sometimes embedded directly in images. Ensuring proper anonymization is critical for compliance. AI helps by:
- Automatically identifying and removing protected health information from DICOM headers
- Detecting and masking burned-in text within images
- Applying consistent redaction rules across large datasets
AI in Image Quality Control
Maintaining image quality and protocol adherence is essential for reliable trial outcomes. AI can proactively identify issues before they impact analysis. It can:
- Detect incomplete, corrupted, or low-quality scans
- Flag deviations from study-specific imaging protocols
- Ensure consistency across sites and timepoints
The Evolving Role of Medical Imaging in Clinical Trials
Clinical trials now span geographies and produce massive imaging datasets, elevating medical imaging from a supporting role to a primary driver of evidence generation, validation, and action. The shift emphasizes intelligent data utilization over sheer volume.
DICOM provides the standardization baseline, but AI advancements are redefining scale: automating image interpretation, quality assurance, and de-identification with unmatched speed and uniformity- tasks previously bogged down by manual processes.
Imaging is also fusing with EDC, ePRO, and other sources, yielding holistic patient profiles. This integration supports adaptive protocols, accelerates decisions, and detects treatment signals sooner. Ultimately, the priority is evolving from data capture to lifecycle-ready insights: always reliable, compliant, and primed for analysis.
Clinion DICOM: Medical Image Repository
Clinion’s DICOM Image Repository streamlines clinical trial imaging with automated upload, AI-driven de-identification in <30 seconds, and secure cloud access. Images are processed and made globally available with minimal manual effort. Integrated directly with the EDC system, each image is linked to subject data and trial records, eliminating silos while ensuring compliance, traceability, and analysis readiness.
See Clinical Trial Imaging in Action
Join our on-demand webinar to explore how Clinion’s Image Repository streamlines imaging workflows, DICOM handling, and centralized data management.
External Reference

Abriti Rai writes on the intersection of AI, automation, and clinical research. At Clinion, she develops content that simplifies complex innovations and highlights how technology is shaping the next generation of data-driven clinical trials.
FAQS
Frequently Asked Questions
Medical imaging is used to objectively measure disease progression and treatment response throughout a clinical trial. It enables researchers to track changes over time, such as tumor size or metabolic activity, using consistent and quantifiable data rather than relying only on clinical observations.
In routine care, imaging is used for diagnosis and patient management. In clinical trials, imaging must follow strict protocols to ensure consistency across sites. The goal is not just interpretation, but generating standardized, comparable data that can support trial endpoints and regulatory submissions.
DICOM (Digital Imaging and Communications in Medicine) is the global standard for storing and sharing medical images. It ensures that images and their associated metadata are structured consistently, making it possible to exchange, analyze, and interpret imaging data reliably across different systems, sites, and devices.
A DICOM file includes both the image itself and detailed metadata. This metadata captures information such as patient identifiers, imaging parameters, device settings, and study details, all of which are essential for accurate interpretation, traceability, and regulatory compliance.
Clinical trials often involve multiple sites using different imaging equipment. Without standardization, differences in image acquisition and formatting can lead to inconsistent data. Standardization ensures that images can be compared reliably across participants, timepoints, and locations.
Imaging biomarkers are measurable features extracted from images, such as tumor size, lesion characteristics, or metabolic activity. They are used to quantify disease status and treatment response, often by comparing changes from a baseline scan across multiple timepoints.
Key challenges include handling large file sizes, managing complex metadata, ensuring accurate anonymization of patient information, and maintaining consistency in interpretation across readers. These challenges often require structured workflows and advanced tools to manage effectively.
AI is used to automate and enhance several aspects of imaging, including image analysis, metadata extraction, workflow management, and quality control. It can process large datasets quickly, reduce variability in interpretation, and identify issues such as protocol deviations or poor-quality scans.
AI can automatically detect and remove protected health information from both DICOM metadata and embedded text within images. This ensures consistent anonymization across large datasets, reduces manual effort, and helps maintain compliance with data privacy regulations.
Clinion uses AI to automate key imaging workflows, including DICOM de-identification in under 30 seconds, metadata handling, and image quality checks. Integrated directly with the EDC system, it ensures imaging data is standardized, linked to subject records, and ready for analysis without manual effort.
Still have questions?
Explore how Clinion AI can accelerate your trial – reach out to our team.
Unlock the Future of Clinical Trials with Clinion.
Cut your trial costs by 35% and accelerate your time-to-market by 30%
Compliance
Fully Compliant with Global Standards


