Insights / Blog / eCOA
eCOA in Clinical Trials: A Complete Guide to Electronic Outcome Assessments
- Abriti Rai
- April 6, 2026

On this Page
- Summary
- Rethinking How Clinical Outcomes Are Captured
- What is eCOA?
- eCOA vs ePRO
- Why eCOA is critical in modern clinical trials
- Types of eCOA
- Key Features of eCOA in Clinical Trials
- eCOA Workflow in Clinical Trials
- Benefits of eCOA in Clinical Trials
- Challenges of Using eCOA in Clinical Trials
- eCOA Migration: Moving from Paper and Legacy Systems
- Best Practices for eCOA Implementation
- Conclusion
- Summary
- Rethinking How Clinical Outcomes Are Captured
- What is eCOA?
- eCOA vs ePRO
- Why eCOA is critical in modern clinical trials
- Types of eCOA
- Key Features of eCOA in Clinical Trials
- eCOA Workflow in Clinical Trials
- Benefits of eCOA in Clinical Trials
- Challenges of Using eCOA in Clinical Trials
- eCOA Migration: Moving from Paper and Legacy Systems
- Best Practices for eCOA Implementation
- Conclusion
Summary
eCOA in clinical trials refers to the electronic capture of clinical outcome data, including patient-reported symptoms, clinician assessments, and performance-based measures, recorded in real time using digital devices rather than paper.
Rethinking How Clinical Outcomes Are Captured
Clinical data is no longer confined to the site. It is now recorded in everyday settings, including at home and between visits, as trials rely more on direct, continuous input.
Electronic Clinical Outcome Assessment, or eCOA, has emerged in response to this change. Instead of relying on paper diaries or delayed site entry, data is captured digitally at the point of experience.
This is not a marginal improvement. Studies have shown that eCOA improves data quality by up to 50 % compared to paper-based methods. The difference comes from real-time capture, fewer transcription steps, and built-in validation checks.
The result is more reliable data that better reflects patient experience.
What is eCOA?
eCOA refers to the digital capture of clinical outcome assessments. These assessments measure how a patient feels, functions, or survives during a clinical trial.
Rather than recording responses on paper, participants, clinicians, or observers enter data through electronic interfaces such as mobile applications, tablets, or web platforms. At its core, eCOA is not a single tool. It is a framework that enables the structured, consistent, and timely collection of outcome data from multiple sources.
eCOA vs ePRO
eCOA and ePRO in clinical trials are closely related, which often leads to them being used interchangeably. However, they refer to different scopes of outcome data collection.
Aspect | eCOA | ePRO |
Scope | Broad category | Subset of eCOA |
Data source | Patients, clinicians, observers, and performance tests | Patients only |
Use case | All outcome assessments | Patient-reported outcomes only |
Why eCOA is critical in modern clinical trials
The adoption of eCOA is not driven by preference. It is driven by necessity. Several factors have made traditional data capture methods less viable:
Increased reliance on patient-reported data
Many endpoints now rely on symptoms, quality of life, and treatment experience reported by patients in real time.
Demand for real-time visibility
Sponsors and monitors require ongoing access to data to identify issues early and respond without delay.
Limitations of paper-based methods
Delayed recording, missing entries, and transcription errors reduce data reliability and increase reconciliation effort.
Regulatory expectations for data integrity
Data must be attributable, contemporaneous, and traceable throughout the study lifecycle.
eCOA addresses these requirements by enabling data to be captured at the time of experience, supported by automated checks, timestamped entries, and centralized access. This shift improves both the integrity and usability of clinical data.
Types of eCOA
eCOA is not a single type of assessment. It brings together multiple outcome measures, each defined by who reports the data and how it is captured. These different inputs are often used together to provide a more complete view of treatment impact.

ePRO (Electronic Patient-Reported Outcomes)
ePRO captures data reported directly by patients, without interpretation by clinicians or others. This includes symptoms, side effects, daily functioning, and quality of life.
It is particularly important for endpoints that cannot be observed externally, such as pain levels or fatigue, and is widely used in studies where patient experience is a primary focus.
eClinRO (Electronic Clinician-Reported Outcomes)
eClinRO includes assessments recorded by investigators or healthcare professionals based on clinical observation and judgment. This may include disease severity scales, physical examination findings, or structured rating tools. These assessments provide a clinical perspective that complements patient-reported data, especially in conditions where medical evaluation is required to interpret outcomes.
eObsRO (Electronic Observer-Reported Outcomes)
eObsRO is used when patients are unable to reliably report their own outcomes. In these cases, caregivers, parents, or other observers record observable signs, behaviors, or daily functioning. This approach is common in pediatric studies, as well as in trials involving cognitive impairment or neurological conditions.
ePerfO (Electronic Performance Outcomes)
ePerfO measures outcomes based on tasks performed by the patient according to standardized instructions. These are objective assessments, such as mobility tests, cognitive exercises, or timed activities. Because they are task-based, ePerfO provides quantifiable data that can reduce subjectivity in outcome measurement.
Each of these assessment types serves a specific role. When used together within an eCOA framework, they allow trials to capture both subjective experience and objective measurement, leading to a more complete and reliable understanding of treatment effects.
Key Features of eCOA in Clinical Trials
The effectiveness of eCOA depends on how well the platform supports consistent, accurate, and user-friendly data capture across all stakeholders. This requires more than digitizing forms. It requires systems designed for real-world use by patients, sites, and sponsors.
Key features include:
Intuitive patient interface
eCOA platforms must be easy to navigate for participants with varying levels of digital familiarity. Clear instructions, simple layouts, and minimal steps reduce the likelihood of incomplete or incorrect entries. This is particularly important in long-duration studies where sustained engagement is required.
Multi-device and BYOD support
Data capture should be accessible across smartphones, tablets, and web-based interfaces. Many studies now follow a bring-your-own-device model, allowing patients to use their personal devices. This reduces logistical burden and supports higher participation rates.
Real-time data capture and availability
Data entered by participants or site staff is immediately available for review. This allows sponsors and monitors to track study progress continuously, identify missing data early, and respond to potential issues without delay.
Offline data capture capability
Continuous internet access cannot be assumed across all geographies. eCOA platforms must allow data to be entered offline and securely synced once connectivity is restored, ensuring no data is lost.
Automated reminders and notifications
Built-in alerts prompt participants to complete scheduled assessments. These reminders play a key role in maintaining compliance, especially in studies that require frequent or time-sensitive data entry.
Data validation and edit checks
Automated checks ensure that data entries are complete, within expected ranges, and logically consistent. This reduces the need for manual queries and minimizes the risk of errors that typically occur with paper-based methods.
Seamless integration with clinical systems
eCOA platforms must integrate with systems such as EDC, RTSM, and data management platforms. This ensures that data flows without duplication, supports centralized monitoring, and maintains a single source of truth across the study.
Audit trails and compliance support
Every data entry and modification should be time-stamped and traceable. This is essential for regulatory compliance and ensures that data integrity is maintained throughout the study lifecycle.
These features are not optional enhancements. They form the foundation for reliable, scalable, and compliant outcome data capture in modern clinical trials.
eCOA Workflow in Clinical Trials
eCOA operates as a continuous data capture workflow, integrated with the broader clinical trial ecosystem. It enables the collection, transmission, and review of outcome data with minimal delay and limited manual intervention.
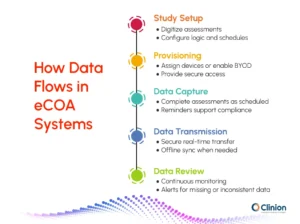
The process typically follows these stages:
Study setup
Clinical outcome assessments are digitized and configured within the system. This includes setting up questionnaires, visit schedules, and logic checks to ensure data is captured in a structured and consistent format.
Provisioning and access
Participants are onboarded onto the platform, either through provisioned devices or a bring-your-own-device approach. Secure access credentials are issued, and initial guidance is provided to ensure correct usage.
Data capture at the point of experience
Participants, clinicians, or observers complete assessments based on predefined schedules. Built-in notifications and reminders help ensure entries are completed on time, supporting higher compliance rates.
Secure data transmission and synchronization
Captured data is transmitted to centralized systems in real time. In cases where connectivity is limited, data can be recorded offline and synchronized once access is restored, ensuring continuity.
Ongoing data review and monitoring
Investigators and sponsors have continuous visibility into incoming data. Automated alerts flag missing, delayed, or inconsistent entries, allowing timely intervention and reducing downstream data queries.
This workflow reduces dependence on manual transcription and delayed entry, while enabling continuous oversight of study data as it is generated.
Benefits of eCOA in Clinical Trials
The benefit of eCOA is reflected across all stakeholders involved in a clinical trial. By enabling direct, real-time data capture, it reduces inefficiencies while improving the quality and usability of outcome data.
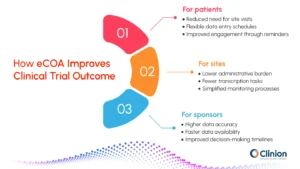
For patients
- Reduced need for frequent site visits, supporting participation from home
- Flexible data entry aligned with daily routines rather than site schedules
- Improved engagement through automated reminders and simple interfaces
- Ability to report symptoms and experiences at the time they occur
For sites
- Reduced administrative burden from manual data entry and paper handling
- Fewer transcription errors and data reconciliation tasks
- Streamlined monitoring with direct access to patient-entered data
- Less time spent on query resolution and follow-ups
For sponsors
- Higher data accuracy through real-time capture and validation checks
- Faster availability of data for review and interim analysis
- Improved visibility into study progress and participant compliance
- More reliable data to support regulatory submissions and decision-making
Across all groups, the outcome is more consistent data capture, reduced operational effort, and better alignment between how data is generated and how it is used.
Challenges of Using eCOA in Clinical Trials
While eCOA improves how outcome data is captured, it also introduces operational considerations that need to be addressed early.
- Patient compliance
Missed or delayed entries can still occur without consistent engagement, particularly in studies with frequent assessments. - Device accessibility
Not all participants may have access to compatible devices or stable connectivity, which can affect participation and data continuity. - Training and onboarding
Both patients and site staff need to be familiar with the system to ensure accurate and consistent data entry. - System integration
Disconnected systems can lead to data silos, increasing the effort required for reconciliation and monitoring.
These challenges are manageable, but they require structured planning and ongoing oversight.
eCOA Migration: Moving from Paper and Legacy Systems
Many studies still rely on paper-based methods or older electronic systems that were not designed for continuous, patient-driven data capture. Transitioning to eCOA is often necessary, but it requires careful coordination.
Key considerations include:
- Assessing existing data collection processes
- Maintaining continuity for ongoing studies
- Aligning stakeholders on updated workflows
- Managing operational change across sites and participants
The objective is not only to replace paper but to ensure that data integrity, consistency, and study timelines are preserved throughout the transition.
Best Practices for eCOA Implementation
Successful eCOA implementation depends on a few consistent principles that support both usability and data reliability.
Prioritize patient usability
Interfaces should be simple, intuitive, and require minimal instruction to reduce entry errors and improve adherence.
Plan training early
Site staff and participants should be comfortable with the system before study initiation to avoid delays.
Ensure interoperability
eCOA platforms should integrate seamlessly with EDC and other clinical systems to support unified data flow.
Pilot before full deployment
Testing the system in a controlled setting helps identify usability or workflow issues early.
Monitor compliance continuously
Ongoing tracking of data entry helps detect gaps early and reduces downstream queries.
These practices support consistent data capture and reduce operational friction during study execution.
Conclusion
eCOA represents a shift toward more reliable and timely capture of clinical outcomes. It supports the growing need for patient-driven data while improving visibility and data quality across the study lifecycle.
As trials continue to extend beyond site-based models, digital outcome capture is becoming a necessary part of study design.
How prepared is your trial infrastructure to support continuous, real-time outcome data capture?
Clinion’s ePRO
Clinion’s ePRO simplifies patient data collection by enabling direct capture of patient-reported outcomes such as symptoms, treatment experience, and quality of life through an intuitive mobile interface. Patients can report data in real time using their own devices, supported by automated reminders for consistent participation. The platform supports multilingual surveys, includes validated questionnaires, and ensures secure, compliant data capture with built-in validation and audit trails. It can be used as a standalone solution or integrated with Clinion EDC for streamlined data flow across studies.

Abriti Rai writes on the intersection of AI, automation, and clinical research. At Clinion, she develops content that simplifies complex innovations and highlights how technology is shaping the next generation of data-driven clinical trials.
FAQS
Frequently Asked Questions
eCOA can be implemented across all trial phases, but it is most impactful when introduced early. Early adoption ensures consistent data capture from the start, reduces the need for mid-study migration, and supports better endpoint evaluation throughout the trial lifecycle.
eCOA captures data at the point of experience, meaning patients report symptoms or events as they occur rather than relying on memory. This significantly reduces recall bias, which is a common issue in paper-based or delayed reporting methods.
eCOA improves endpoint accuracy by standardizing how data is collected and validated. With real-time checks and structured formats, it ensures that endpoint data is complete, consistent, and aligned with protocol-defined requirements.
eCOA platforms use automated reminders, scheduling logic, and timestamped entries to ensure assessments are completed within defined windows. This is critical for endpoints that depend on precise timing, such as dosing diaries or symptom tracking.
Yes, eCOA platforms are highly configurable. Questionnaires, assessment frequency, logic checks, and workflows can be tailored to specific therapeutic areas such as oncology, CNS, or rare diseases, as well as different study designs.
eCOA systems support multiple languages and culturally validated questionnaires, allowing consistent data collection across regions. This ensures that global trials maintain data standardization while accommodating diverse patient populations.
Missed entries are flagged automatically within the system. Alerts notify site staff or sponsors, enabling timely follow-up. Some platforms also allow controlled back-entry within defined limits to maintain data continuity without compromising integrity.
eCOA platforms use encryption, role-based access controls, and secure data transmission protocols. Combined with audit trails and compliance with regulatory standards, these measures ensure patient data is protected throughout the study.
eCOA integrates with systems like EDC, RTSM, and CTMS to enable seamless data flow. This reduces duplication, supports centralized monitoring, and ensures that all stakeholders work from a unified dataset.
Yes, modern eCOA platforms are designed with simple interfaces, clear instructions, and minimal navigation steps. Combined with training and support, they can be effectively used even by participants with limited digital experience.
By capturing data directly from patients in their daily environments, eCOA provides insights that reflect real-world conditions. This makes it a valuable tool for generating high-quality real-world evidence alongside clinical trial data.
Key metrics include patient compliance rates, data completeness, query rates, time to data availability, and reduction in protocol deviations. These indicators help assess both operational efficiency and data quality improvements.
Still have questions?
Explore how Clinion AI can accelerate your trial – reach out to our team.
Unlock the Future of Clinical Trials with Clinion.
Cut your trial costs by 35% and accelerate your time-to-market by 30%
Compliance
Fully Compliant with Global Standards


