Insights / Blog / Decentralized Trials
Decentralized Clinical Trials: How Research Is Moving Beyond Traditional Sites
- Abriti Rai
- April 21, 2026

On this Page
- Summary
- What Are Decentralized Clinical Trials
- How Decentralized Clinical Trials Differ From Traditional Clinical Trials
- How Decentralized Clinical Trials Work in Practice
- What Types of Clinical Trials Can Be Decentralized
- Why Decentralized Clinical Trials Are Gaining Importance
- Technologies Enabling Decentralized Clinical Trials
- Active vs Passive Data Collection in Decentralized Clinical Trials
- Roles, Responsibilities, and Patient Engagement in Decentralized Clinical Trials
- Benefits of Decentralized Clinical Trials
- AI Across the Decentralized Clinical Trial Lifecycle
- Challenges in Decentralized Clinical Trials
- Regulatory Considerations for Decentralized Clinical Trials
- Conclusion: The Evolution of Decentralized Clinical Trials
- External References
- Summary
- What Are Decentralized Clinical Trials
- How Decentralized Clinical Trials Differ From Traditional Clinical Trials
- How Decentralized Clinical Trials Work in Practice
- What Types of Clinical Trials Can Be Decentralized
- Why Decentralized Clinical Trials Are Gaining Importance
- Technologies Enabling Decentralized Clinical Trials
- Active vs Passive Data Collection in Decentralized Clinical Trials
- Roles, Responsibilities, and Patient Engagement in Decentralized Clinical Trials
- Benefits of Decentralized Clinical Trials
- AI Across the Decentralized Clinical Trial Lifecycle
- Challenges in Decentralized Clinical Trials
- Regulatory Considerations for Decentralized Clinical Trials
- Conclusion: The Evolution of Decentralized Clinical Trials
- External References
Summary
Decentralized clinical trials (DCTs) are studies that use digital tools and remote processes to reduce or eliminate the need for in-person site visits. They enable patients to participate from home while enabling continuous data collection through connected systems and digital platforms.
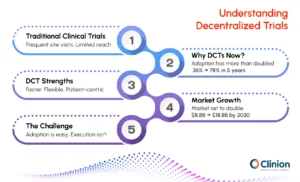
Traditional clinical trials rely on physical sites and in-person visits to collect and validate data. While this model has supported clinical research for decades, it can also create limitations around patient access and operational efficiency.
Decentralized clinical trials introduce an alternative approach where patient participation is supported through digital infrastructure rather than being anchored to a single physical site.
What Are Decentralized Clinical Trials
Decentralized clinical trials are best understood as a model where clinical research activities are distributed across digital systems, local healthcare providers, and patient environments instead of being concentrated at a central site.
Data in decentralized clinical trials is captured through digital platforms and connected devices. This allows study teams to collect information continuously rather than only during scheduled site visits.
A study conducted in Singapore reported a 97% retention rate in a fully decentralized trial, highlighting how reduced patient burden can positively influence participation continuity.
Decentralized clinical trials shift the model from being site-centric to being more patient-centric. The focus moves from where the trial happens to how the patient participates.
Key Characteristics of Decentralized Clinical Trials
Decentralized clinical trials are defined by a few core characteristics that distinguish them from traditional approaches:
- Participation can happen remotely through virtual visits or telemedicine
- Data is captured digitally rather than through manual site entry
- Dependence on centralized trial sites is significantly reduced
- Study designs can combine remote and site-based elements depending on requirements
These characteristics can be combined in different ways depending on the study design.
How Decentralized Clinical Trials Differ From Traditional Clinical Trials
To understand the value of decentralized clinical trials, it helps to compare them directly with traditional models.
Table: Traditional vs Decentralized Clinical Trials
Aspect | Traditional Clinical Trials | Decentralized Clinical Trials |
Patient participation | Site-based | Remote or hybrid |
Data collection | On-site | Digital and continuous |
Accessibility | Limited to site locations | Broader geographic reach |
Monitoring | In-person visits | Remote monitoring |
In traditional clinical trials, patients are required to travel to designated sites for most study activities. This can create barriers, especially for participants who live far from research centers or have limited mobility.
Decentralized clinical trials reduce this dependency by enabling remote interactions and digital data capture. This allows participation to extend beyond geographic constraints while maintaining study oversight.
How Decentralized Clinical Trials Work in Practice
Decentralized clinical trials are not defined by a single structure. Instead, they bring together different tools and processes to support remote participation.
Patient Journey in Decentralized Clinical Trials
The patient journey in a decentralized clinical trial often begins with digital recruitment. Participants may be identified through online platforms, databases, or referrals. Screening can be conducted remotely using digital questionnaires or virtual consultations.
Once enrolled, patients complete the consent process electronically. This often includes guided interfaces that help them understand study requirements before signing.
During the study, data is collected through mobile applications, wearable devices, or scheduled virtual visits. Follow-ups and monitoring are also conducted remotely, reducing the need for frequent site visits.
Types of Decentralized Clinical Trials
There are two primary ways decentralized clinical trials are implemented.
Fully decentralized clinical trials conduct all activities remotely, including recruitment and monitoring. These are more common in studies that do not require complex procedures.
Hybrid decentralized clinical trials combine remote activities with occasional in-person visits. This approach is often used when certain assessments must be conducted on-site while others can be managed remotely.
Understanding how decentralized clinical trials work raises a more practical question: can this approach apply to every type of study?
What Types of Clinical Trials Can Be Decentralized
Decentralized clinical trials are not limited to one category, but they are not suitable for every study in the same way. The level of decentralization depends on how the trial is designed and what it requires from patients.
Some types of clinical trials can be conducted mostly remotely. Others still need a combination of site visits and remote participation.
Trials That Can Be Mostly Decentralized
These trials do not rely heavily on in-person procedures and can be managed using digital tools and remote interactions.
- Observational studies that track patient outcomes over time (Example: monitoring long COVID symptoms through a mobile app).
- Long-term follow-up studies after treatment (Example: tracking recovery after a cardiac procedure through periodic virtual check-ins).
- Chronic condition studies with routine monitoring (Example: diabetes trials using glucose monitoring devices and patient-reported data).
- Studies based on patient-reported outcomes (Example: mental health trials where patients log symptoms and responses digitally).
Trials That Typically Use a Hybrid Decentralized Approach
These trials require some level of physical interaction, but not for every step of the study. Here, decentralization reduces the number of visits, but does not eliminate them.
- Trials that include lab tests or imaging at specific intervals (Example: oncology trials where scans are done at a site, but follow-ups happen remotely).
- Studies where certain assessments must be performed by clinicians (Example: neurological trials requiring in-person cognitive assessments).
- Trials involving treatments that require occasional administration at a site (Example: infusion therapies with remote monitoring between visits).
- Studies combining remote tracking with periodic evaluations (Example: cardiovascular trials using wearables with scheduled in-clinic check-ups).
Trials That Are Less Suitable for Full Decentralization
Some trials depend heavily on controlled environments and close supervision. These trials are typically conducted at sites, with limited decentralization.
- Early-phase trials that require intensive monitoring (Example: first-in-human drug studies conducted in controlled clinical units).
- Studies involving complex procedures or interventions (Example: surgical trials or device implantation studies).
- Trials where investigational products need strict handling (Example: therapies requiring cold-chain storage and controlled administration).
- High-risk studies requiring continuous clinical oversight (Example: trials with significant safety monitoring requirements).
See how Clinion supported trial continuity during COVID-19 with decentralized solutions.
Why Decentralized Clinical Trials Are Gaining Importance
The growing interest in decentralized clinical trials is driven by both operational challenges and changing expectations across the industry.
One key factor is the need to improve recruitment timelines. Traditional trials often struggle to enroll participants quickly, especially when limited to specific locations. Expanding participation beyond physical sites helps address this issue. There is also an increasing focus on improving diversity in clinical trials. Decentralized approaches make it easier to reach participants from different geographic and demographic backgrounds.
At the same time, advances in digital health technologies have made remote data collection more reliable. Patients are also becoming more comfortable with digital interactions, which supports broader adoption of decentralized models.
Technologies Enabling Decentralized Clinical Trials
Technology is what makes decentralized clinical trials possible. It allows studies to move beyond site boundaries while still maintaining control over how data is captured and managed across systems.
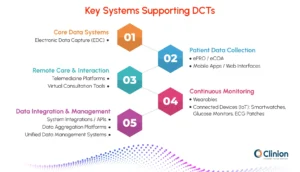
At the core are Electronic Data Capture (EDC) systems, which act as the foundation for structuring clinical data. In decentralized trials, EDC platforms integrate with external data sources so that remotely collected information flows into a single system for review.
ePRO (electronic Patient-Reported Outcomes) and eCOA (electronic Clinical Outcome Assessments) platforms enable patients to directly report symptoms, experiences, and outcomes through mobile apps or web interfaces. This reduces reliance on site visits and improves the accuracy of patient-reported data by capturing it in real time rather than retrospectively.
Telemedicine technologies support virtual interactions between patients and investigators. These tools enable remote consultations, follow-ups, and even certain assessments, helping maintain continuity in patient engagement without requiring physical site visits.
Wearables and connected devices play a critical role in continuous monitoring. Devices such as smartwatches, glucose monitors, and ECG patches can capture high-frequency data like heart rate, activity levels, sleep patterns, or vital signs. This provides a more comprehensive and objective view of patient health compared to periodic site-based measurements.
Beyond individual tools, the real impact comes from how these technologies work together. Integrations between EDC, ePRO/eCOA, telemedicine platforms, and devices ensure that data from multiple sources is consolidated, traceable, and ready for analysis.
Active vs Passive Data Collection in Decentralized Clinical Trials
Data in decentralized trials is collected through two primary approaches, each serving a distinct purpose.
Active data collection
Active data collection depends on direct input from patients or study staff.
This includes reporting symptoms or adverse events, completing questionnaires or study diaries through digital tools, and recording medication use or daily health patterns.
This type of data captures the patient’s experience and provides context that cannot be measured through devices alone. However, it relies on consistent patient engagement and timely reporting.
Passive data collection
Passive data collection happens automatically through connected devices.
This includes continuous tracking of physiological signals such as heart rate, activity levels, or sleep patterns, as well as automated health data capture without manual input.
This approach reduces patient effort and enables uninterrupted data collection over time. It also helps identify patterns that may not be visible through periodic reporting.
How these approaches work together
In practice, decentralized clinical trials combine both active and passive data collection methods. Active inputs provide context around patient experience and reported outcomes, while passive monitoring captures continuous physiological changes.
Together, they create a more complete and real-world view of patient health that traditional site-based models often cannot achieve.
Roles, Responsibilities, and Patient Engagement in Decentralized Clinical Trials
Sponsors are responsible for designing decentralized clinical trials so that study processes work effectively across both remote and site-based settings. They also oversee vendors and ensure that technology platforms remain compliant and reliable.
Investigators continue to play a central role in patient safety and oversight. Even in virtual settings, they review study data, support clinical decisions, and ensure that patient care standards are maintained throughout the study.
Technology and service providers form the operational backbone of decentralized clinical trials. They enable data capture across systems, maintain platform stability, and ensure accurate information flow between stakeholders.
Benefits of Decentralized Clinical Trials
Decentralized clinical trials create value across the clinical research ecosystem, but the impact varies depending on the stakeholder. Each group experiences different improvements in access, efficiency, and operational flow based on how the study is designed.
Overview of Key Benefits
Stakeholder | Key Benefits |
Patients | Reduced travel burden, greater flexibility in participation, improved access to clinical research |
Sponsors & CROs | Faster recruitment, broader patient reach, more consistent real-world data collection |
Clinical Sites | Reduced administrative workload, improved focus on complex procedures, and better resource utilization |
Benefits for Patients
For patients, decentralized clinical trials make participation significantly easier by reducing the need for frequent site visits. This is especially valuable for individuals who live far from clinical centers or have mobility or time constraints.
They also improve how patients interact with the study, allowing participation to fit more naturally into daily life rather than requiring major schedule adjustments.
Key patient benefits include:
- Reduced need for travel to clinical sites
- Greater flexibility in completing study activities
- Improved access to clinical trials regardless of location
- Easier participation for patients with mobility or time limitations
Benefits for Sponsors and CROs
For sponsors and CROs, decentralized clinical trials improve the speed and reach of patient recruitment. By removing geographic limitations, studies can access a broader and more diverse patient population.
They also enable more continuous data collection through digital tools, reducing reliance on isolated site visits and improving data flow throughout the study.
In addition, decentralized models can strengthen the quality of real-world data by capturing patient information in everyday settings.
Key sponsor and CRO benefits include:
- Faster patient recruitment and enrollment timelines
- Access to a wider and more diverse patient population
- Continuous data collection through digital platforms
- Improved real-world relevance of study data
Benefits for Clinical Sites
For clinical sites, decentralized clinical trials reduce the burden of routine trial activities. This allows site staff to spend more time on complex procedures that require clinical expertise.
It also improves how site resources are used, particularly in studies with high patient volumes or long follow-up periods.
Key clinical site benefits include:
- Reduced workload from routine trial activities
- More focus on complex clinical and protocol-driven tasks
- Improved allocation of site resources
Better operational efficiency in high-volume studies
AI Across the Decentralized Clinical Trial Lifecycle
AI in decentralized clinical trials operates as a continuous intelligence layer rather than a standalone tool. It connects different stages of the clinical trial lifecycle by enabling the interpretation and coordination of data across systems as it is generated.
- In early-stage activities, AI helps identify relevant patient populations by analyzing clinical and real-world datasets
- During trial conduct, it interprets incoming data from multiple sources, such as patient inputs, site systems, and connected devices
- In later stages, it supports the generation of insights that extend beyond the study period into real-world evidence
Beyond the active trial phase, AI extends its role into post-trial analysis by enabling the interpretation of long-term outcomes and supporting the generation of real-world evidence. This allows data collected during the study to contribute to broader clinical understanding beyond the trial itself. Across all stages, AI functions as a connecting layer that enables a continuous flow of information between recruitment, execution, and post-trial analysis, ensuring data is interpreted in a unified manner
Challenges in Decentralized Clinical Trials
While decentralized clinical trials offer clear advantages, they also introduce operational, technological, and regulatory complexities that need to be carefully managed.
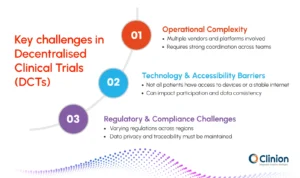
Operational complexity
Managing decentralized clinical trials often involves coordinating multiple vendors, platforms, and service providers across different regions. Unlike traditional trials, where activities are centralized at study sites, decentralized models distribute responsibilities across several stakeholders. This increases the need for strong coordination and clear communication between all parties involved in the study.
Technology and accessibility barriers
Technology plays a central role in decentralized clinical trials, but it also introduces variability in access and usability. Not all patients have equal access to smartphones, wearable devices, or stable internet connections. This can create gaps in participation and may affect how consistently data is collected across the study population.
Regulatory and compliance considerations
Decentralized clinical trials must comply with regulatory requirements across different regions while also maintaining data privacy and security standards. This becomes more complex when data is collected through multiple digital systems and transferred across platforms.
Ensuring consistency in compliance practices and maintaining traceability of data are key challenges in this model.
Regulatory Considerations for Decentralized Clinical Trials
Regulatory frameworks for decentralized clinical trials (DCTs) continue to evolve, yet core principles remain unchanged: patient safety, data integrity, and full traceability must be upheld regardless of study location or methodology.
Data privacy and security
- Compliance with GDPR, HIPAA, and applicable regional data protection regulations.
- Secure storage, transmission, and access controls for patient data.
- Explicit informed consent processes tailored for digital tools and remote collection.
System compliance
- Validation of EDC, ePRO, telemedicine platforms, and wearable devices.
- Adherence to 21 CFR Part 11, GxP standards, and ALCOA+ principles.
- Reliable, audit-ready electronic records with user access controls.
Audit trails and traceability
- End-to-end traceability across hybrid and remote systems.
- Time-stamped, version-controlled records for all data changes.
- Comprehensive audit trails documenting access, modifications, and approvals.
Vendor oversight
- Rigorous compliance assessments for all third-party providers.
- Clearly defined responsibilities via formal contracts and SLAs.
- Continuous monitoring and performance audits of vendor operations.
Conclusion: The Evolution of Decentralized Clinical Trials
Decentralized clinical trials represent a modern approach to clinical research that combines digital technologies with distributed study execution models. Rather than relying on a single physical site, they enable data collection and patient participation across multiple environments.
As digital infrastructure continues to mature, decentralized models are becoming more integrated into mainstream clinical development. Hybrid approaches are increasingly common, combining remote capabilities with traditional site-based processes where needed.
The growing role of AI, connected devices, and interoperable systems is further strengthening the foundation of decentralized research by improving how data is captured, connected, and interpreted.
The future of clinical trials is expected to be defined by more distributed, data-connected, and patient-aligned models of research execution.
How Clinion Enables Decentralised Clinical Trials
Clinion supports decentralized clinical trials by making it easier for both patients and study teams to participate and manage studies within a single platform. With EDC and ePRO built together, patients can report outcomes directly through simple digital interfaces, while site teams can review the same data in real time without switching systems. This reduces complexity in how data is collected and used across the study. By supporting both remote participation and site-based inputs in one place, Clinion helps studies run more smoothly while keeping data organized, accessible, and aligned with regulatory requirements.
External References
Decentralized Clinical Trials Market to Reach $18.8 Billion by 2030 as Digital Health Infrastructure Reshapes Drug Development
Decentralized Clinical Trials Market Size & Share | DCT Industry Trends, 2031.
Decentralized clinical trials are better for the participants and for the planet: the case study of a double-blind randomized controlled trial in Singapore (PROMOTE study) - PubMed.
The Introduction of AI Into Decentralized Clinical Trials: Preparing for a Paradigm Shift - PMC

Abriti Rai writes on the intersection of AI, automation, and clinical research. At Clinion, she develops content that simplifies complex innovations and highlights how technology is shaping the next generation of data-driven clinical trials.
FAQS
Frequently Asked Questions
Decentralized clinical trials are studies that use digital tools and remote processes to enable patient participation without relying entirely on physical trial sites. Data is collected through connected systems, devices, and digital platforms.
They combine digital recruitment, electronic consent, remote data collection, and ongoing monitoring to manage study activities. Depending on the design, trials may be fully remote or include selected in-person visits.
Traditional trials depend on site visits for most study activities, whereas decentralized trials distribute these activities across digital platforms, local providers, and patient environments.
Yes, patient safety is maintained through continuous data monitoring, investigator oversight, and predefined safety protocols, even when participation happens remotely.
Common technologies include EDC systems, ePRO and eCOA platforms, wearable devices, and other digital tools that support remote data capture and study management.
Data quality is maintained through standardized systems, real-time data capture, audit trails, and validation processes that ensure accuracy and traceability.
Not always. Some trials are fully remote, while others follow a hybrid model where only specific procedures require site visits.
They reduce the need for travel and allow patients to participate from their own environment, making it easier to enroll and remain in studies.
In some cases, yes. However, many studies still use a hybrid approach where certain activities, such as imaging or lab tests, are conducted at clinical sites.
Depending on the study, investigational products may be shipped directly to patients, administered at local healthcare facilities, or managed at clinical sites based on safety and handling requirements.
Local providers may support activities such as lab testing, physical assessments, or treatment administration, helping reduce the need for patients to travel to central trial sites.
Still have questions?
Explore how Clinion AI can accelerate your trial – reach out to our team.
Unlock the Future of Clinical Trials with Clinion.
Cut your trial costs by 35% and accelerate your time-to-market by 30%
Compliance
Fully Compliant with Global Standards


